 Two postgraduate students, each working independently on their own research project, had to compare the corrosion rates of magnesium alloys in salt water measured by several techniques. Both obtained puzzling results, which can be summarized as follows:
Four techniques yielded corrosion rates on the order of 1–5 mm/year. The fifth technique — measurement of gas evolution during corrosion — gave a rate of about 30 mm/year, which is unusually high. Moreover, the average corrosion rate measured by gas evolution during the day was significantly lower — roughly four times — than that measured at night. The results were readily reproducible, so we had to find an explanation.
Having excluded ghosts, security officers, and other supernatural factors, we turned to temperature, atmospheric pressure, and humidity, all of which differ between day and night. Since temperature was easiest to measure, we started there and found that the day-to-night difference could be as large as 10°C.
Normally, corrosion is faster at higher temperatures — so the activation energy argument worked in the wrong direction. A temperature-dependent change in corrosion mechanism was possible in principle, but we set that aside for later. Viscosity could also be ruled out: at night the solution is more viscous, which should reduce gas evolution, not increase it. We had no basis for estimating the effect of humidity and assumed it was negligible.
Pressure remained. Fortunately, a group in a neighboring lab had been logging atmospheric pressure twice daily. Their data showed almost no day-to-night difference.
In an ideal system, the manometric pressure of gas collected in a burette should equal atmospheric pressure exactly. In reality there is some thermal inertia: a measurement taken early in the morning might still reflect the night temperature of the electrolyte. For an ideal gas (pV = nRT) this effect could be estimated, and it amounted to roughly 5% — far less than what we observed. Besides, thermal inertia might explain the first morning measurement, but not readings taken hours later.
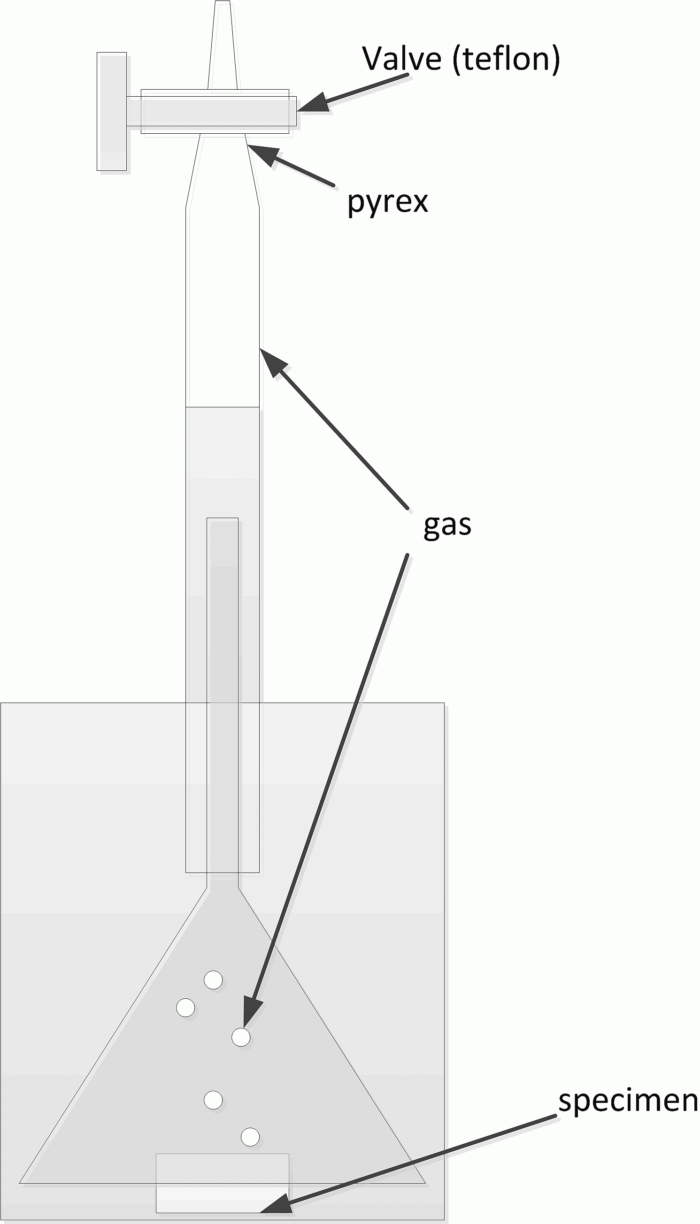
At this point we were stuck. Being stuck, as it turns out, is very useful: it forces you to go back and check everything. We did, and soon found that our burettes were leaking. All of them. We replaced them, verified the new ones for leaks, and the gas evolution rates immediately fell into agreement with all the other techniques.
We still wanted to understand why leaky burettes always read higher at night than during the day. The answer lay in the thermal expansion coefficients: Pyrex (the burette body) has a coefficient of 4×10⁻⁶ K⁻¹, while Teflon (the stopcock) has 135×10⁻⁶ K⁻¹ — more than thirty times larger. Our estimate showed that a clearance of 0.1 mm between the Pyrex and Teflon parts during the day would grow to approximately 0.13 mm at night, as the Teflon contracted far more than the glass. The leak was worse at night, so more gas escaped, so the apparent corrosion rate was paradoxically lower — which is exactly what we observed.
Two postgraduate students, each working independently on their own research project, had to compare the corrosion rates of magnesium alloys in salt water measured by several techniques. Both obtained puzzling results, which can be summarized as follows:
Four techniques yielded corrosion rates on the order of 1–5 mm/year. The fifth technique — measurement of gas evolution during corrosion — gave a rate of about 30 mm/year, which is unusually high. Moreover, the average corrosion rate measured by gas evolution during the day was significantly lower — roughly four times — than that measured at night. The results were readily reproducible, so we had to find an explanation.
Having excluded ghosts, security officers, and other supernatural factors, we turned to temperature, atmospheric pressure, and humidity, all of which differ between day and night. Since temperature was easiest to measure, we started there and found that the day-to-night difference could be as large as 10°C.
Normally, corrosion is faster at higher temperatures — so the activation energy argument worked in the wrong direction. A temperature-dependent change in corrosion mechanism was possible in principle, but we set that aside for later. Viscosity could also be ruled out: at night the solution is more viscous, which should reduce gas evolution, not increase it. We had no basis for estimating the effect of humidity and assumed it was negligible.
Pressure remained. Fortunately, a group in a neighboring lab had been logging atmospheric pressure twice daily. Their data showed almost no day-to-night difference.
In an ideal system, the manometric pressure of gas collected in a burette should equal atmospheric pressure exactly. In reality there is some thermal inertia: a measurement taken early in the morning might still reflect the night temperature of the electrolyte. For an ideal gas (pV = nRT) this effect could be estimated, and it amounted to roughly 5% — far less than what we observed. Besides, thermal inertia might explain the first morning measurement, but not readings taken hours later.
At this point we were stuck. Being stuck, as it turns out, is very useful: it forces you to go back and check everything. We did, and soon found that our burettes were leaking. All of them. We replaced them, verified the new ones for leaks, and the gas evolution rates immediately fell into agreement with all the other techniques.
We still wanted to understand why leaky burettes always read higher at night than during the day. The answer lay in the thermal expansion coefficients: Pyrex (the burette body) has a coefficient of 4×10⁻⁶ K⁻¹, while Teflon (the stopcock) has 135×10⁻⁶ K⁻¹ — more than thirty times larger. Our estimate showed that a clearance of 0.1 mm between the Pyrex and Teflon parts during the day would grow to approximately 0.13 mm at night, as the Teflon contracted far more than the glass. The leak was worse at night, so more gas escaped, so the apparent corrosion rate was paradoxically lower — which is exactly what we observed.
