(Working notes)
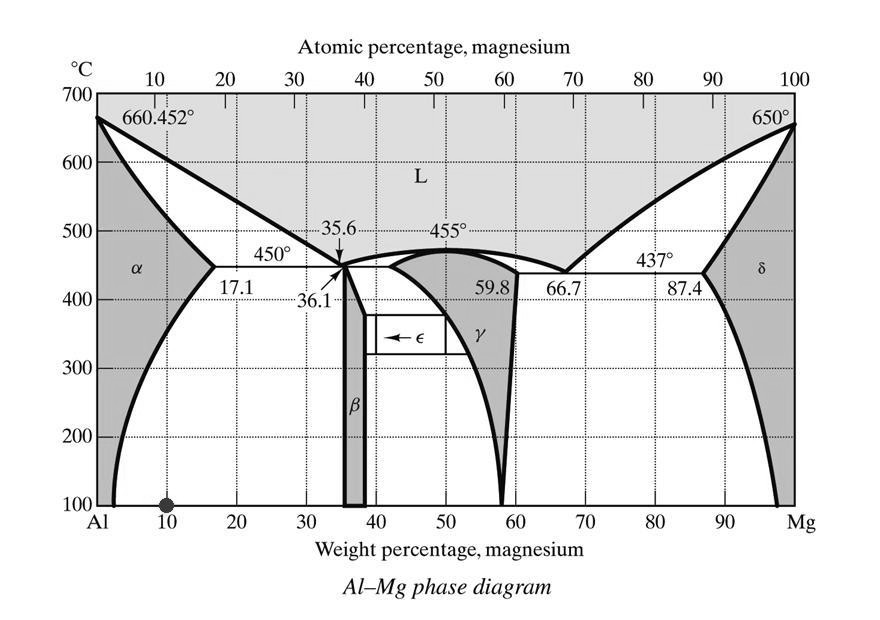
Virtually any Mg alloy containing aluminium consists of two main phases (the influence of minor additives, which can be significant, is ignored here):
• α-phase: essentially pure magnesium (though it may contain up to ~17 at.% Al in solid solution)
• β-phase: Mg₁₇Al₁₂ (calculated composition: 55.7 wt% Mg)
A note on nomenclature: strictly speaking, according to the equilibrium Al–Mg phase diagram, the phase commonly called β should be referred to as γ, since a congruent compound exists at approximately 50 at.% Al. The “true” β-phase has a composition of approximately 35–37 at.% Al and the nominal stoichiometry Al₈Mg₅. This phase is occasionally mentioned in the literature but is never referred to as β. We follow the mainstream convention and use β for Mg₁₇Al₁₂ throughout.
The free corrosion potentials of the relevant phases are as follows ¹˒²:
| phase | , V vs. SHE |
|---|---|
| -phase | - 1.651 |
| -phase () | - 1.201 |
| - 12.252 | |
| Both intermetallic phases are strongly cathodic with respect to the α-phase, and would therefore be expected to accelerate its oxidation. In practice, however, the corrosion behavior depends strongly on microstructural factors ³: |
• If the volume fraction of the β-phase is small, it acts as a local cathode and accelerates corrosion of the surrounding α-phase. When the volume fraction is sufficiently large, the β-phase forms a more or less continuous network and can act as a protective barrier, retarding corrosion.
• For the same volume fraction of β-phase, an alloy with finer grain structure corrodes more slowly than one with coarser grains. Finer α-phase grains are enveloped by a nearly continuous thin network of β-phase, which provides effective protection. This is why die-cast alloys typically corrode more slowly than sand-cast alloys.
• The periphery of an ingot corrodes more slowly than the core — sometimes by a factor of ten — because peripheral grains are finer and better protected by the β-phase network.
References 1. O. Lunder, K. Nisancioglu, R. S. Hansen, SAE Technical Paper Series, 1993, 930755. 2. K. Mutombo, M. Du Toit, “Corrosion fatigue behaviour of aluminium 5083-H111 welded using gas metal arc welding,” in Arc Welding, InTech Open Access, 2011, pp. 177–218. 3. G. Song, A. Atrens, “Understanding Magnesium Corrosion — A Framework for Improved Alloy Performance,” Advanced Engineering Materials, 2003, 5(12), 837–858.
